 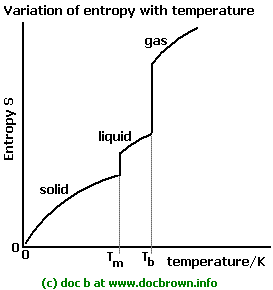 
heat engine or heat pump) exchanging heat with external thermal reservoirs and undergoing a thermodynamic cycle, the following inequality holds. Note: Thermodynamically, all naturally occurring spontaneous reactions are irreversible. So to compute "the" enthalpy $H$ of say $\ce$! So if those two things are equal, and one of them is a state function, then the other one must be a state function too! So $\Delta H$ is also a state function. So can we say in other words that: 'state variable' is something that we take as independent variable, while 'state function' is something that depends on previously selected 'state variables' where this dependence is given in the equation of state for the particular thermodynamic system. Entropy is a state function because it depends only on the arrangement (as a statistical average) of the constituents of the system at the given values of the thermodynamic variables (T,P,V etc), not how the arrangement was obtained. The Clausius theorem (1855), also known as the Clausius inequality, states that for a thermodynamic system (e.g. Entropy is a state function since it depends not only on the start and end states, but also on the entropy change between two states, which is integrating tiny entropy change along a reversible route. 
5.3: Unlike heat, Entropy is a State Function is shared under a not declared license and was authored, remixed, and/or curated. When a gas expands into a vacuum, its entropy increases because the increased volume allows for greater atomic or molecular disorder. Clearly entropy is a state function while qrev q r e v is not. As with any other state function, the change in entropy is defined as the difference between the entropies of the final and initial states: S S f S i. More broadly we define a spontaneous process as one that occurs in the. Both paths had the same change in entropy. Like enthalpy, entropy is a thermodynamic term that is also a state function. We could also a standard state where all the atoms in a sample are separated into pure chemical elements. We took two different paths to get start and end at the same points. We could say the standard state is one where all electrons and all protons and neutrons are infinitely separated.but that's possibly only useful for nuclear chemistry. For example, the standard state might be all electrons and all nuclei infinitely separated from each other (this standard is commonly used in quantum chemistry).īut we could also specify other standard states. Entropy is a state function whose magnitude depends only on the. This is as far as you can deduce using thermodynamics of an ideal gas only.) For a non-ideal gas, the. S Nk ln VTcv N S N k ln V T c v N ( cv c v is the specific heat of the gas at constant volume, and is a gas-dependent constant. So really even in defining $H$, there is some built-in standard state that you are comparing the state of interest to. Entropy is defined as a quantitative measure of disorder or randomness in a system. PV NkT P V N k T (which makes F(P, V, T) PV NkT F ( P, V, T) P V N k T ) and. To define topological entropy in the approximation approach, it suffices to replace the 2-norm defined by the invariant state by the. Everyone agrees $H$ is a state function, but energy (and thus enthalpy) has no built in zero-scale. If the process in question is irreversible, then computing $\int \delta Q/T$ will yield a result which is strictly less than $\Delta S$.I generally agree with DavePhD's answer but I also want to give the opposite view (!). afrykman 12 years ago The integral was just the mathematical way to write the heat, Q, in terms of the volume, V, because that's what he had the proof in terms of from the last video. Thermodynamicists know that and have adopted a specific tactic. The proof offered by Clausius is based on the assumption 'A general cycle can be divided into small Carnot cycles' which is almost obviously wrong. Put differently, given any two states $1$ and $2$, the entropy difference can be computed by finding any reversible process which takes $1$ to $2$ and calculating $\int_1^2 \delta Q/T$ for that process. Yet the premise 'Entropy is a state function' has no serious justification. I think the way you write the two conditions may be misleading. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
